A) These amide bonds are not stable.
B) There is partial double bond character between the carbonyl carbon and the nitrogen atom.
C) The oxygen atom is hydrogen-bonded with the nitrogen atom.
D) The side-chains can form hydrogen bonds.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major organic product for the following reaction? 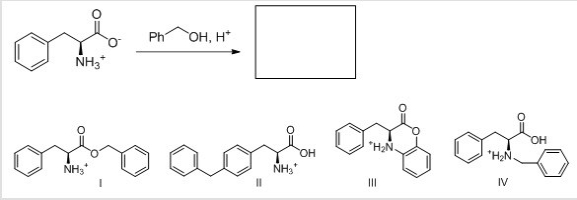
A) I
B) II
C) III
D) IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the name given to the pH at which the concentration of the zwitterionic form of an amino acid is at a maximum concentration?
A) Electric point
B) Dipolar point
C) Neutral point
D) Isoelectric point
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why is the s-trans form of an amide bond more stable?
A) There is no s-cis form in an amide bond.
B) The oxygen is hydrogen-bonded with the nitrogen atom.
C) The nitrogen is able to be sp2-hybridized.
D) The more sterically bulky groups are farther apart.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the separation of a racemic mixture into its component enantiomers called?
A) Diastereomers
B) Meso
C) Resolution
D) Neutralization
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Reaction of a polypeptide,composed of 12 amino acids,with carboxypeptidase A releases Met (C-terminal amino acid) .The polypeptide undergoes partial hydrolysis to give 12 groups of peptides.Use the groups of overlapping amino acids to determine the proper sequence of this polypeptide.Note: Since these lists of peptides are separated by commas,they are not necessarily in the proper sequence. Ser,Lys,Trp-5.Met,Ala,Gly- 9.Lys,Ser Gly,His,Ala-6.Ser,Lys,Val -10.Glu,His,Val Glu,Val,Ser-7.Glu,His-11.Trp,Leu,Glu Leu,Glu,Ser-8.Leu,Lys,Trp-12.Ala,Met
A) Met-Ala-Gly-His-Glu-Val-Ser-Lys-Trp-Leu-Glu-Ser
B) Met-Ala-Gly-Glu-His-Ser-Val-Lys-Trp-Leu-Glu-Ser
C) Ser-Glu-Leu-Trp-Lys-Ser-Val-Glu-His-Gly-Ala-Met
D) Ser-Lys-Leu-Trp-Lys-Ser-Val-His-Glu-Gly-Ala-Met
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 46 of 46
Related Exams