A) 15.6 kJ
B) 10.2 kJ
C) 5.4 kJ
D) 3.2 kJ
E) 1.6 kJ
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The phase diagram for xenon has a solid-liquid curve with a positive slope. Which of the following is true?
A) Solid xenon has a higher density than liquid xenon.
B) Solid xenon has the same density as liquid xenon.
C) The phase diagram cannot be used to predict which phase of xenon is denser.
D) Freezing xenon is an endothermic process.
E) None of these choices are correct.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is true about kinetic energy, Ek, and potential energy, Ep, when ethyl alcohol at 40°C is compared with ethyl alcohol at 20°C?
A) Ek(40°C) < Ek(20°C) ; Ep(40°C) ≈ Ep(20°C)
B) Ek(40°C) > Ek(20°C) ; Ep(40°C) ≈ Ep(20°C)
C) Ep(40°C) < Ep(20°C) ; Ek(40°C) ≈ Ek(20°C)
D) Ep(40°C) > Ep(20°C) ; Ek(40°C) ≈ Ek(20°C)
E) Ep(40°C) > Ep(20°C) ; Ek(40°C) > Ek(20°C)
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following liquid substances would you expect to have the lowest surface tension?
A) Pb
B) CH3OCH3
C) HOCH2CH2OH
D) H2O
E) CH3CH2OH
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Octane is a component of fuel used in internal combustion engines. The dominant intermolecular forces in octane are
A) dipole-dipole forces.
B) London dispersion forces.
C) hydrogen bonding.
D) covalent bonds.
E) carbon-hydrogen bonds.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following should have the highest boiling point?
A) CF4
B) CCl4
C) CBr4
D) CI4
E) CH4
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about the packing of monatomic solids with different unit cells is incorrect?
A) The coordination number of atoms in hcp and fcc structures is 12.
B) The coordination number of atoms in simple cubic structures is 6.
C) The coordination number of atoms in bcc structures is 8.
D) A bcc structure has a higher packing efficiency than a simple cubic structure.
E) A bcc structure has a higher packing efficiency than a fcc structure.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Acetic acid has a heat of fusion of 10.8 kJ/mol and a heat of vaporization of 24.3 kJ/mol. Estimate the value for the heat of sublimation.
A) 35.1 kJ/mol
B) 13.5 kJ/mol
C) -13.5 kJ/mol
D) -35.1 kJ/mol
E) None of these choices are correct
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Polonium crystallizes in the simple cubic lattice. What is the coordination number for Po?
A) 3
B) 4
C) 6
D) 8
E) 12
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
A single water molecule can participate in at most two hydrogen bonds at any instant.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A sample of octane in equilibrium with its vapor in a closed 1.0-L container has a vapor pressure of 50.0 torr at 45°C. The container's volume is increased to 2.0 L at constant temperature and the liquid/vapor equilibrium is reestablished. What is the vapor pressure?
A) > 50.0 torr
B) 50.0 torr
C) 25.0 torr
D) The mass of the octane vapor is needed to calculate the vapor pressure.
E) The external pressure is needed to calculate the vapor pressure.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
In metals, the conduction bands and valence bands of the molecular orbitals are separated by a large energy gap.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
All gases can be liquefied at room temperature simply by increasing the pressure on the gas.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
The phase diagram of a substance shows the energy changes associated with changes of state.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following pairs of molecules can form hydrogen bonds between them?
A) HCl and HI
B) CH3OH and NH3
C) CH4 and H2O
D) SO2 and CH2O
E) H2 and O2
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following should have the highest surface tension at a given temperature?
A) CH4
B) CF4
C) CCl4
D) CBr4
E) CI4
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Diethyl ether, used as a solvent for extraction of organic compounds from aqueous solutions, has a high vapor pressure which makes it a potential fire hazard in laboratories in which it is used. How much energy is released when 100.0 g is cooled from 53.0°C to 10.0°C? Boiling point: 34.5°C Heat of vaporization: 351 J/g Specific heat capacity, (CH3) 2O(l) : 3.74 J/(g·K) Specific heat capacity, (CH3) 2O(g) : 2.35 J/(g·K)
A) 10.1 kJ
B) 13.1 kJ
C) 16.1 kJ
D) 45.2 kJ
E) 48.6 kJ
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Liquid ammonia (boiling point = -33.4°C) can be used as a refrigerant and heat transfer fluid. How much energy is needed to heat 25.0 g of NH3(l) from -65.0°C to -12.0°C?
Specific heat capacity, NH3(l) : 4.7 J/(g·K)
Specific heat capacity, NH3(g) : 2.2 J/(g·K)
Heat of vaporization: 23.5 kJ/mol
Molar mass, 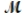 : 17.0 g/mol
: 17.0 g/mol
A) 5.5 kJ
B) 6.3 kJ
C) 39 kJ
D) 340 kJ
E) 590 kJ
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following atoms should have the greatest polarizability?
A) F
B) Br
C) Po
D) Pb
E) He
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When identical particles pack in a simple cubic lattice, there is/are ____ particle(s) per unit cell.
A) 1
B) 2
C) 3
D) 4
E) 8
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 90
Related Exams