A) Cr
B) Cr(II)
C) Cr(III)
D) Cr(IV)
E) Cr(VI)
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following ions is least likely to form colored compounds?
A) Mn2+
B) Cr5+
C) Sc3+
D) Fe3+
E) Co2+
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Write the formula for sodium tetracyanonickelate(II) .
A) Na[Ni(CN) 4]
B) Na[Ni(CN) 4]2
C) Na2[Ni(CN) 4]
D) Na4[Ni(CN) 4]
E) None of these choices is correct.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following ions could exist in only the high-spin state in an octahedral complex?
A) Cr2+
B) Mn4+
C) Fe3+
D) Co3+
E) Ni2+
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The ground state electronic configuration of Cr2+ is:
A) [Ar]4s13d5
B) [Ar]4s23d4
C) [Ar]3d4
D) [Ar]4s13d3
E) [Ar]4s23d2
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the highest possible oxidation state for molybdenum, Mo?
A) +2
B) +4
C) +6
D) +8
E) None of these choices is correct.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to Valence Bond theory, in the square planar Ni(CN) 42¯ complex ion, the orbital hybridization pattern is:
A) sp3
B) dsp2
C) d2sp
D) d2sp3
E) None of these choices is correct.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the spectrochemical series, which one of the following ligands has the strongest field?
A) H2O
B) CN¯
C) NH3
D) OH¯
E) Cl¯
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following should be the strongest reducing agent?
A) Fe
B) Ru
C) Os
D) Re
E) Cu
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The most common oxidation state for ions of the transition elements is:
A) +2
B) +3
C) +4
D) +5
E) +6
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Mercury(II) compounds are assimilated into the food chain because
A) they are ionic substances that are concentrated in cell tissue.
B) they complex with compounds in the blood which carries them to the muscle tissue of organisms.
C) they are incorporated into bone structure.
D) they are very soluble in water and easily ingested.
E) they are nonpolar substances that are concentrated in fatty tissues as they move up the food chain.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following ions is most likely to form colored compounds?
A) Sc3+
B) Cu+
C) Zn2+
D) Cr3+
E) Ca2+
G) None of the above
Correct Answer

verified
Correct Answer
verified
True/False
All atoms of the first transition series of elements have the ground state electronic configuration [Ar]4s23dx, where x is an integer from 1 to 10.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the formation of a transition metal complex, the central metal atom or ion acts as
A) an Arrhenius acid.
B) a Brønsted-Lowry acid.
C) a Brønsted-Lowry base.
D) a Lewis acid.
E) a Lewis base.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many unpaired electrons are there in the Fe3+ ion?
A) 5
B) 4
C) 3
D) 2
E) 1
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following octahedral complexes should have the largest crystal field splitting energy, ?
A) [Cr(H2O) 6]3+
B) [Cr(SCN) 6]3¯
C) [Cr(NH3) 6]3+
D) [Cr(CN) 6]3¯
E) [Cr(en) 3]3+ (en = ethylenediamine)
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Of the 3d transition series of elements, scandium has the greatest atomic radius.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following octahedral complex structures, each involving ethylene diamine and two different, unidentate ligands X and Y. 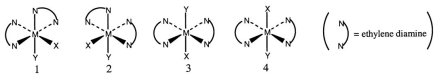 Which one, if any, of the following is a pair of optical isomers?
Which one, if any, of the following is a pair of optical isomers?
A) 1 and 2
B) 1 and 3
C) 1 and 4
D) 3 and 4
E) None of these choices is correct.
G) None of the above
Correct Answer

verified
Correct Answer
verified
True/False
The Cu2+ ion has 1 unpaired electron.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A characteristic of ligands is that
A) they are Lewis acids.
B) they are Lewis bases.
C) they are ions.
D) they are electron pair acceptors.
E) they are Brønsted-Lowry acids.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 92
Related Exams