A) The frequency of collisions of molecules with the walls is increased.
B) The average velocity of the molecules is lowered.
C) The temperature of the sample is altered.
D) Half of the molecules are replaced by an equal number of molecules of a gas with a different molecular weight.
E) The total number of molecules is altered.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
"The volume of an ideal gas is directly proportional to the number of moles of the gas at constant temperature and pressure" is a statement of __________ Law.
A) Charles's
B) Boyle's
C) Amontons's
D) Avogadro's
E) Dalton's
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A carbon dioxide sample weighing 44.0 g occupies 32.68 L at 65°C and 645 torr. What is its volume at STP?
A) 22.4 L
B) 31.1 L
C) 34.3 L
D) 35.2 L
E) 47.7 L
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The air pressure in a volleyball is 75 psi. What is this pressure in torr?
A) 520 torr
B) 562 torr
C) 3900 torr
D) 7600 torr
E) 75000 torr
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An unknown liquid is vaporized in a 273-mL flask by immersion in a water bath at 99°C. The barometric pressure is 753 torr. If the mass of the vapor retained in the flask is 1.362 g, what is its molar mass?
A) 20.4 g/mol
B) 40.9 g/mol
C) 112 g/mol
D) 154 g/mol
E) 184 g/mol
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
"The total pressure in a mixture of unreacting gases is equal to the sum of the partial pressures of the individual gases" is a statement of __________ Law.
A) Charles's
B) Graham's
C) Boyle's
D) Avogadro's
E) Dalton's
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the root-mean-square speed of methane, CH4 (g) , at 78°C.
A) 23 m/s
B) 350 m/s
C) 550 m/s
D) 667 m/s
E) 740 m/s
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
When a closed-ended manometer is used for pressure measurements, and the closed end is under vacuum, the level of manometer liquid in the closed arm can never be lower than that in the other arm.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
At a temperature of absolute zero, the volume of an ideal gas is zero.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the gas with the largest root-mean-square molecular speed at 25°C.
A) NH 3
B) CO
C) H 2
D) SF 6
E) All the gases have the same root-mean-square molecular speed at 25°C.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Mercury is 13.6 times as dense as liquid water. What would be the reading of a water-filled barometer at normal atmospheric pressure, 760. mmHg?
A) 1.03 × 10 3 torr
B) 1.03 × 10 3 Pa
C) 1.03 × 10 3 mm height of water column
D) 1.03 × 10 3 cm height of water column
E) 13.6 atm
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Linolenic acid (C18H30O2) can be hydrogenated to stearic acid by reacting it with hydrogen gas according to the equation: C18H30O2 + 3H2 → C18H36O2 What volume of hydrogen gas, measured at STP, is required to react with 10.5 g of linolenic acid in this reaction?
A) 2.53 L
B) 1.69 L
C) 1.27 L
D) 845 mL
E) 422 mL
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The pressure of hydrogen sulfide gas in a container is 35,650 Pa. What is this pressure in torr?
A) 46.91 torr
B) 267.4 torr
C) 351.8 torr
D) 3612 torr
E) 27090 torr
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the lines on the figure below is the best representation of the relationship between the volume of a gas and its absolute temperature, other factors remaining constant? 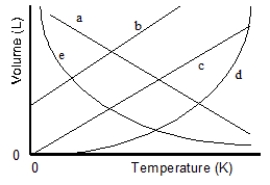
A) a
B) b
C) c
D) d
E) e
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the lines on the figure below is the best representation of the relationship between the volume of a gas and its pressure, other factors remaining constant? 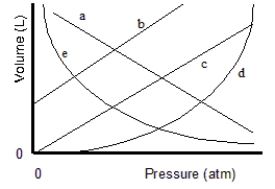
A) a
B) b
C) c
D) d
E) e
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A compound composed of carbon, hydrogen, and chlorine effuses through a pinhole 0.411 times as fast as neon. Select the correct molecular formula for the compound.
A) CHCl 3
B) CH 2Cl 2
C) C 2H 2Cl 2
D) C 2H 3Cl
E) CCl 4
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the gas with the highest average kinetic energy per mole at 298 K.
A) O 2
B) CO 2
C) H 2O
D) H 2
E) All have the same average kinetic energy.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
True/False
For real gases, PV > nRT, always.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A sample of propane, a component of LP gas, has a volume of 35.3 L at 315 K and 922 torr. What is its volume at STP?
A) 25.2 L
B) 30.6 L
C) 33.6 L
D) 37.1 L
E) 49.2 L
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Arrange the following gases in order of increasing rate of effusion. C2H6 Ar HCl PH3
A) Ar < HCl < PH 3 < C 2H 6
B) C 2H 6 < PH 3 < HCl < Ar
C) Ar < PH 3 < C 2H 6 < HCl
D) C 2H 6 < HCl < PH 3 < Ar
E) Ar < PH 3< HCl < C 2H 6
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 98
Related Exams