A) drawing (a)
B) drawing (b)
C) drawing (c)
D) drawing (d)
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the reaction shown below,what is the relationship between the rate of formation of Br2 and the rate of reaction of Br- over the same time period? ClO2-(aq) + 4 Br-(aq) + 4 H+(aq) → Cl-(aq) + 2 Br2(aq) +2 H2O(l)
A) rate of formation of Br2 = 1/2 × rate of reaction of Br-
B) rate of formation of Br2 = 1/4 × rate of reaction of Br-
C) rate of formation of Br2 = 2 × rate of reaction of Br-
D) Rate of formation of Br2 = 2 × rate of reaction of Br-
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which statement below regarding the half-life of a second-order reaction is true?
A) Each half-life is half as long as the preceding one.
B) Each half-life is twice as long as the preceding one.
C) Each half-life is four times as long as the preceding one.
D) The length of the half-life remains unchanged throughout the course of the reaction.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The decomposition of ozone in the stratosphere can occur by the following two-step mechanism: Br + O3 → BrO + O2 BrO + O → Br + O2 Which species is a catalyst in this mechanism?
A) Br
B) BrO
C) O
D) O3
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Hydrogen iodide decomposes at 800 K via a second-order process to produce hydrogen and iodine according to the following chemical equation. 2 HI(g) → H2(g) + I2(g) At 800 K it takes 142 seconds for the initial concentration of HI to decrease from 6.75 × 10-2 M to 3.50 × 10-2 M.What is the rate constant for the reaction at this temperature?
A) 5) 12 × 10-4 M-1s-1
B) 9) 69 × 10-2 M-1s-1
C) 10.3 M-1s-1
D) 1) 95 × 103 M-1s-1
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the first-order reaction,2 N2O(g) → 2 N2(g) + O2(g) ,what is the concentration of N2O after 3 half-lives if 0.25 mol of N2O is initially placed into a 1.00-L reaction vessel?
A) 1) 6 × 10- 2 M
B) 3) 1 × 10- 2 M
C) 6) 2 × 10- 2 M
D) 1) 2 × 10- 1 M
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
 -What is the activation energy for the formation of ozone?
-What is the activation energy for the formation of ozone?
A) 14 kJ
B) 392 kJ
C) 406 kJ
D) none of these
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The reaction for the decomposition of dinitrogen monoxide gas to form oxygen radicals is: N2O(g) N2(g) + O(g) .If the rate constant is 3.04 × 10-2 s-1 and the frequency factor is 8) 00 × 1011 s-1,what is the activation energy for the first-order reaction at 700°C?
A) 0) 262 kJ/mol
B) 38.2 kJ/mol
C) 180 kJ/mol
D) 250 kJ/mol
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following set of data was obtained by the method of initial rates for the reaction:
(H3C) 3CBr + OH- → (H3C) 3COH + Br-
What is the value of the rate constant,k? 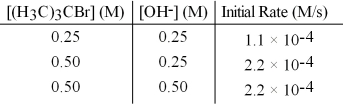
A) 8) 8 × 10-4 s-1
B) 4) 4 × 10-4 s-1
C) 1) 8 × 10-4 s-1
D) none of these
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A mechanism for a naturally occurring reaction that destroys ozone is: Step 1: O3(g) + HO(g) → HO2(g) + O2(g) Step 2: HO2(g) + O(g) → HO(g) + O2(g) What is the molecularity of the overall reaction?
A) unimolecular
B) bimolecular
C) none of these because molecularity is the difference of the exponents in the rate law
D) none of these because molecularity only refers to elementary steps
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
The reaction below is first order in H2 and one-half order in Br2.The rate law for this reaction is ________. H2(g)+ Br2(g)→ 2 HBr(g)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A gaseous reaction occurs by a two-step mechanism,shown below. Step 1: AX +Y2 ⇌ AXY2 fast Step 2: AXY2 + AX → 2 AXY slow Including concentration of only reactants and products,what is the rate law for this reaction?
A) Rate = k[AX][Y2]
B) Rate = k[AXY2]/[AX][Y2]
C) Rate = k[AX]2[Y2]
D) Rate = k[AXY]2/[AXY2][AX]
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Methanol can be produced by the following reaction:
CO(g) + 2 H2(g) → CH3OH(g) .
How is the rate of disappearance of hydrogen gas related to the rate of appearance of methanol? 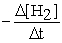 = ?
= ?
A) ![]()
B) ![]()
C) ![]()
D) none of these
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The first-order isomerization reaction: cyclopropane → propene,has a rate constant of 1) 10 × 10-4s-1 at 470°C and an activation energy of 264 kJ/mol.What is the temperature of the reaction when the rate constant is equal to 4.36 × 10-3s-1?
A) 126°C
B) 411°C
C) 510°C
D) 540°C
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A concentration-time study of the gas phase reaction 2 A3 → 3 A2 produced the data in the table below. Time (s) [A3] (M) [A2] (M) 0 4) 00 × 10-4 0 10 2.00 × 10-4 3.00 × 10-4 20 1.00 × 10-4 4.50 × 10-4 30 5.00 × 10-5 ? What is the concentration of A2 after 30 seconds?
A) 5) 00 × 10-4 M
B) 5) 25 × 10-4 M
C) 5) 50 × 10-4 M
D) 6) 00 × 10-4 M
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the zeroth-order reaction: C → products,-Δ[C]/Δt = k,which of the following graphs would be expected to give a straight line?
A) [C] vs.t
B) ln[C] vs.t
C) 1/[C] vs.t
D) [C]2 vs.t
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A plot of 1/[BrO-] vs time is linear for the reaction: 3 BrO-(aq) → BrO3-(aq) + 2 Br-(aq) What is the order of the reaction with respect to the hypobromite ion,BrO-?
A) 0
B) 1
C) 2
D) 3
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
A reaction has the rate law Rate = k[NO]2[H2].If the concentration of NO is reduced by half and the concentration of H2 is quadrupled,the rate of reaction will ________ (increase,decrease,not change).
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Cerium(IV) ion reacts with thallium(I) ion in a one-step reaction shown below: 2 Ce4+(aq) + Tl+(aq) → 2 Ce3+(aq) + Tl3+(aq) . If the rate law is: Rate = k[Ce4+]2[Tl+],what is the overall order of the reaction?
A) first
B) second
C) third
D) fourth
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Hydrogen peroxide decomposes to water and oxygen according to the reaction below:
2 H2O2(aq) → 2 H2O(l) + O2(g)
In the presence of large excesses of I- ion,the following set of data is obtained.What is the average rate of disappearance of H2O2(aq) in M/s in the first 45.0 seconds of the reaction if 1.00 L of H2O2 reacts at 25°C and 1.00 atm pressure? 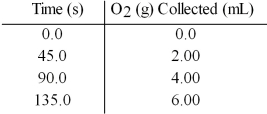
A) 9) 09 × 10-7 M/s
B) 3) 63 × 10-6 M/s
C) 4) 33 × 10-5 M/s
D) 1) 64 × 10-4 M/s
F) None of the above
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 206
Related Exams