Filters
Question type
A) 5.86
B) 6.30 108
C) 1.59 10-9
D) 5.47 10-21
E) 2.18 10-27
F) C) and E)
G) B) and C)
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Question 52
Multiple Choice
Which of the following is always true for an endothermic process?
A) q sys > 0, Ssurr < 0
B) qsys < 0, Ssurr > 0
C) qsys < 0, Ssurr < 0
D) qsys > 0, Ssurr > 0
E) w < 0
F) B) and C)
G) A) and E)
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Question 53
Multiple Choice
Calculate S° for the combustion of propane.
C3H8(g) + 5O2(g) 3CO2(g) + 4H2O(g)
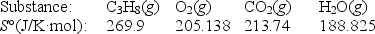
A) -100.9 J/K
B) -72.5 J/K
C) 72.5 J/K
D) 100.9 J/K
E) 877.5 J/K
F) A) and B)
G) B) and E)
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Question 54
Multiple Choice
Calculate G° for the reaction of ammonia with fluorine.
2NH3(g) + 5F2(g) N2F4(g) + 6HF(g)
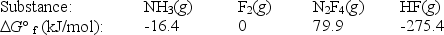
A) 179.1 kJ
B) -179.1 kJ
C) 1539.7 kJ
D) -1539.7 kJ
E) None of these choices is correct.
F) A) and B)
G) B) and E)
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Showing 81 - 84 of 84
Related Exams