A) Na+(aq) + NO3-(aq) NaNO3(aq)
B) 3Co2+(aq) + NO3-(aq) + Na+(aq) + 2PO43-(aq) Co3(PO4) 2(s) + NaNO3(aq)
C) 3Co2+(aq) + 6NO3-(aq) + 6Na+(aq) + 2PO43-(aq) Co3(PO4) 2(s) + 6NaNO3(aq)
D) 3Co2+(aq) + 2PO43-(aq) Co3(PO4) 2(s)
E) None of the above is the correct net ionic equation.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the precipitate that forms when aqueous lead(II) nitrate reacts with aqueous sodium sulfate.
A) NaNO3
B) Na2NO3
C) PbSO4
D) Pb2SO4
E) PbS
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following substances, when dissolved in water at equal molar concentrations, will give the solution with the lowest electrical conductivity?
A) CaCl2
B) HNO3
C) NH3
D) C6H12O6 (glucose)
E) CO2
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the precipitate that forms when the following reactants are mixed. Mg(CH3COO) 2(aq) + LiOH(aq)
A) LiCH3COO
B) Li(CH3COO) 2
C) MgOH
D) Mg(OH) 2
E) CH3OH
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many milliliters of 1.58 M HCl are needed to react completely with 23.2 g of NaHCO3 ( 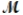 = 84.02 g/mol) ? HCl(aq) + NaHCO3(s) NaCl(s) + H2O(l) + CO2(g)
= 84.02 g/mol) ? HCl(aq) + NaHCO3(s) NaCl(s) + H2O(l) + CO2(g)
A) 638 mL
B) 572 mL
C) 536 mL
D) 276 mL
E) 175 mL
G) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
In a redox reaction, the oxidizing agent undergoes loss of electrons.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is most soluble in water?
A) benzene, C6H6
B) potassium nitrate, KNO3
C) carbon tetrachloride, CCl4
D) hexane, C6H14
E) ethane, C2H4
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Essay
An aqueous solution of lead nitrate, Pb(NO3)2, is mixed with one of sodium chromate, Na2CrO4, resulting in the formation of a precipitate of lead chromate. Write a balanced net ionic equation for this precipitation reaction, showing all phases.
Correct Answer

verified
Pb2+(aq) + C...View Answer
Show Answer
Correct Answer
verified
View Answer
True/False
The correct method for preparing one liter of a 1.0 M solution of X is to dissolve exactly one mole of X in exactly one liter of water.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the classification for the following reaction. 2Ag+(aq) + Zn(s) 2Ag(s) + Zn2+(aq)
A) displacement
B) decomposition
C) combination
D) precipitation
E) acid-base
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the classification for the following reaction. 2NaCl(l) 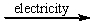 2Na(l) + Cl2(g)
2Na(l) + Cl2(g)
A) acid-base
B) precipitation
C) combination
D) displacement
E) decomposition
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the net ionic equation for the reaction between sodium chloride and mercury(I) nitrate. 2NaCl(aq) + Hg2(NO3) 2(aq) NaNO3(aq) + Hg2Cl2(s)
A) Na+(aq) + NO3-(aq) NaNO3(aq)
B) Hg22+(aq) + 2Cl-(aq) Hg2Cl2(s)
C) NaCl(aq) Na+(aq) + Cl-(aq)
D) Hg2(NO3) 2(aq) Hg22+(aq) + 2NO3-(aq)
E) Hg22+(aq) Hg2(s)
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Essay
For each of the following species, write down, next to the formula, the oxidation number of the indicated atom. a. P in H2PO2- b. S in Na2S2O3 c. C in CH2O
Correct Answer

verified
Correct Answer
verified
Essay
In each of the following cases, write down the oxidation number of the indicated atom. a. P in P4 b. C in C2H6 c. S in H2SO4 d. Mn in MnO4- e. S in S4O62- f. P in Na3PO4
Correct Answer

verified
a. 0
b. -3...View Answer
Show Answer
Correct Answer
verified
b. -3...
View Answer
Multiple Choice
Which one of the following substances is the best electrolyte?
A) CO
B) CH3Cl
C) CH4
D) C2H5OH
E) HCl
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the classification for the following reaction. 2H2O2(aq) 2H2O(l) + O2(g)
A) combination
B) decomposition
C) displacement
D) acid-base
E) none of the above
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a weak base?
A) NH3
B) Ca(OH) 2
C) Ba(OH) 2
D) NaOH
E) CH3COOH
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
Sulfuric acid (H2SO4) reacts with potassium hydroxide (KOH) as follows. H2SO4(aq) + 2KOH(aq) K2SO4(aq) + 2H2O(l) Calculate the volume of 0.100 M sulfuric acid required to neutralize 25.0 mL of 0.0821 M KOH. Show all your work.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many moles of ions are released when 0.27 mol of cobalt(II) chloride, CoCl2, is dissolved in water?
A) 0.81 mol
B) 0.54 mol
C) 0.27 mol
D) 0.18 mol
E) 0.090 mol
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the precipitate that forms when the following reactants are mixed. Na2CO3(aq) + BaCl2(aq)
A) Ba2CO3
B) BaCO3
C) NaCl
D) NaCl2
E) BaO
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 124
Related Exams